Federal regulators are discussing artificial wombs, a crucial step for doctor-researchers who want to pursue the first clinical trials for humans.
FDA advisers are meeting this week on artificial womb and artificial placenta technology. The device would not replace a human womb, but may work as a bridge for very premature babies or what scientists are calling "neonates" after they are born and as they grow.
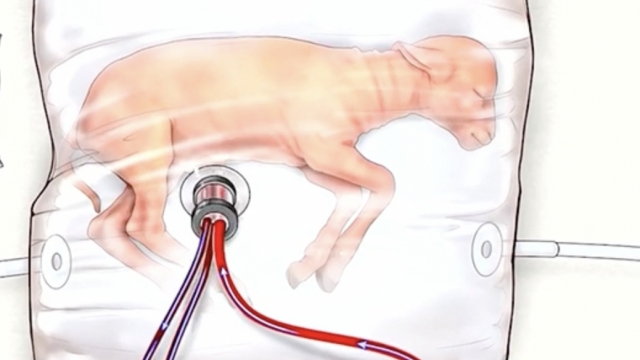
Videos shared by researchers from the University of Michigan and Children's Hospital of Philadelphia show what the technology looks like. The artificial wombs may include a bag with synthetic amniotic fluid and tubes connected to the umbilical cord. They remove harmful carbon dioxide from the blood and bring oxygen and nutrients in.
Over the past six years, researchers say, these artificial wombs have helped brain, lung and gut development in preterm lambs and fetal pigs.
"We've been able to rapidly convert pig fetuses that are human in size from placental support to circuit support with physiologic blood flows," said Dr. Alan Flake, the director at the center for fetal research at the Children's Hospital of Philadelphia.
FDA advisers will discuss potential guidelines for the first studies moving from preterm animals to humans. Questions include, how would moving a patient to an artificial womb work? What would qualify a patient for an artificial womb? What is the ethical way to design the first studies?
Preterm births are those 37 weeks and younger. Cases 32 weeks or younger are considered very to extremely premature. CDC data shows 1 in 10 U.S. babies are born premature. Sixty-five percent of all infant deaths are premature babies.
It will still be several months, if not years, before any clinical trials in humans. Individual research hospitals would also have their own ethics and science reviews before any human tests could take place.
Trending stories at Scrippsnews.com